Curated OER
Quantum Mechanics
Mostly what you will find here is a history of quantum mechanics. Influential scientists are introduced (along with some of their recorded quotes), the progression of atomic models is reviewed, and finally experssions and equations are...
Curated OER
Atoms and Elements - Electron Arrangement and Periodic Law
Delve into the world of quantum mechanics by viewing this PowerPoint. Chemistry novices learn about electron energy and shells, and they practice listing electron configuration for different atoms. Because this presentation provides...
Concord Consortium
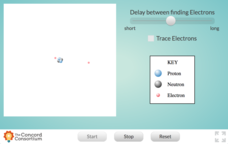
Structure of an Atom
Feeling a little uncertain about your materials for teaching the quantum mechanical model of the atom? Here is an interactive that will help! Chemistry and physics scholars alike will benefit from a simple resource that illustrates the...
Curated OER
Physics and the Quantum Mechanical Model
In this physics activity, students review vocabulary terms and key equations associated with the quantum mechanical model. Students apply the quantum theory to explain the photoelectric effect. This activity has 5 true or false, 12...
Curated OER
Modern Physics, The Quantum
In this physics worksheet, learners examine the relationship between the discrete and continuous spectrum alongside other concepts for the 9 questions.
PhET
Rutherford Scattering
Rutherford performed his famous experiment in 1907 with Marsden, showing the true atomic structure. This interactive simulation compares Rutherford's historical experiment to the Plum pudding model of the atom. In both models, alpha...
Curated OER
Location of Electrons
In this location of electrons worksheet, students read about the quantum mechanical model of the atom and the location of electrons. They complete a table given eleven elements with the sublevel notation, the Bohr notation and the...
Science Geek
Electrons in Atoms
Electrons could never be Bohring! The presentation covers where you find electrons in an atom. It begins with the Bohr Model, then moves on to the Heisenberg Principle and orbital shapes. It concludes with the concept of electron spin...
Curated OER
Indinite Potential Well
Students use experiments such as electron diffraction that show that particle have a wavelike nature. When they are fired through a thin slit, rather than scattering like hard spheres they interfere like waves. Students see that the...
Curated OER
Physicists or Philosophers?
Trace the sequential process of the developing theories of atomic structure in the early 20th century, show, in historical development, how scientists "know" things, how experiments are set up and how interpretations are drawn from them,...
Curated OER
The Flame and the Atom:
High schoolers investigate the structure of atoms. Students read information about the Rutherford model, the Bohr model, and the Quantum Mechanical model examining each for its scientific validity. They watch a PowerPoint presentation of...
Curated OER
Atomic Structure and Quantum Theory
Students are introduced to the structure of an atom and Dalton's atomic theory through a short video and mini-lecture. They also take a look at the weight of mass and become familiar with the idea of quantum theory
Teach Engineering
Electromagnetic Radiation
How can nanoparticles be used in the battle against skin cancer. Class members take on the question as they gather information about electromagnetic radiation, specifically ultraviolet radiation. Pupils learn about the mathematical...
CK-12 Foundation
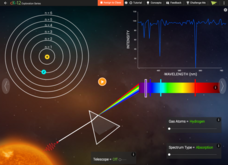
Atomic Colors
Stars are too hot to visit, so how do we know what different stars are made of? An enlightening simulation uses a spectrum graph to show the various electron emission and absorption reactions. Scholars experiment with both helium and...
Curated OER
Grating Spectrometer
Students calculate the Balmer series. In this physics lesson, students observe hydrogen lamp spectra using spectrometers. They calculate wavelength and compare them with their theoretical calculations.
Curated OER
Quantum Mechanics and Atomic Physics
Students will discuss the de Brogile Hypothesis and state the circumstances under which the wave nature of matter is observed. They will also calculate the wavelengths of matter waves.
National Institute of Open Schooling
Atomic Structure
Learners explain historical findings such as Rutherford and Bohr's contributions, explain wave particle duality, and formulate Heinsenberg's uncertainty principle. They also draw s, p, and d orbitals, explain more historical findings,...
Curated OER
Historical Development of a Scientific Idea
Ninth graders examine how scientist's contribute to atomic theory. In this development of a scientific idea lesson plan students work in groups and research the development of the atomic model.
Curated OER
The Day the Atom Died (Grade K-1)
Students investigate what is wrong with the Rutherford atomic model. In this chemistry lesson, students answer questions about the atomic model after watching a video. They draw conclusions from the video and discuss how the atomic model...
Curated OER
The Story of the Atom
In this atom activity, students read about the model of the atom developed by Neils Bohr. Students complete 2 short answer questions based on what they read.
Curated OER
Lesson- Infinite Potential Well
Student create a numerical model which integrates Schrodinger's equation in 1 dimension. They discuss the properties of the solutions which match the boundary conditions. Student discusss any limitations if there are any in there model.
SRI International
Science of Water
Water is crucial to survival. Scholars gain an appreciation for water by reading about it, learning about its atomic properties, and investigating its properties through six stations in a lab activity.
Cornell University
Sometimes You Behave Like a WAVE, Sometimes You Don't!
Electromagnetic radiation behaves like both a wave and a particle. Help classes explore this concept through a lab investigation. Young scientists create optical interference patterns on a glass slide using a carbon layer. They analyze...
Illustrative Mathematics
Accuracy of Carbon 14 Dating II
The scientific issue of carbon-14 dating and exponential decay gets a statistics-based treatment in this problem. The class starts with a basic investigation of carbon content, but then branches out to questions of accuracy and ranges of...