Curated OER
The Nature of Molecules
Even though the questions on this worksheet pertain to general chemistry, it was created for an AP biology course. It is crucial for biology learners to grasp these concepts as a foundation for biochemical processes that they will...
Curated OER
Of Electron Bondage
Students create models of ionic and covalent electronic bonds to demonstrate their understanding of these chemistry concepts. They explain how both ionic bonds and covalent bonds work so that younger students can understand.
Curated OER
Ionic and Covalent Bonds
Ionic and covalent bonds are the focus of this chemistry activity, which provides students with eighteen key terms to use in a fill-in-the-blank activity. Additionally, students are prompted to write the number of atoms in four given...
Curated OER
The Chemistry of Life
After studying the different aspects of atoms and their reactivity, pupils will find this summary PowerPoint useful for review. Some of the slides are informative with labeled diagrams; others give important vocabulary. Teachers may want...
Curated OER
IPC Review
Students discuss the difference between atoms and elements. They analyze how compounds are formed. Students discuss the difference between covalent bonds, hydrogen bonds, and ionic bonds. They review bonding with diagrams, "Atoms or...
Curated OER
Candy Compounds
In this compounds worksheet, learners are given a bag of colored gumdrops that represent specific atoms. They construct ionic and covalent bonds with gumdrops using the key provided and the compounds to build. They fill in a chart with...
Curated OER
The World of Molecules
Students investigate molecules through 2 demonstrations and writing formulas for molecules given the oxidation states of the atoms. In this molecules lesson plan, students observe iron filings being separated from salt and pepper using a...
Curated OER
Intermolecular Forces Worksheet
In this intermolecular forces worksheet, students answer 4 questions about the forces between atoms within molecules and compounds. Students rank the boiling points of compounds based on their intermolecular forces and they explain...
Curated OER
More Chemical Bonding
For this chemical bonding worksheet, learners review the three types of bonds including ionic, covalent and polyatomic compounds. They identify 8 compounds as ionic, covalent or polyatomic and they make 4 ionic compounds and they draw 5...
Curated OER
More Chemical Bonding
In this chemical bonding learning exercise, students review the three types of compounds: ionic, covalent, and polyatomic. Students practice drawing the covalent bonds of given compounds. This learning exercise has 5 drawings and 13 fill...
Curated OER
Ionic Bonding
In this ionic bonding worksheet, students review the characteristics of ionic bonding, draw Lewis dot diagrams for elements, and write the chemical formula for ionic compounds. This worksheet has 8 drawings, 14 fill in the blank, and 6...
Curated OER
The Chemical Context of Life
In this chemistry worksheet, students fill in the chart for question one. The chart is filled out examining the mass, protons, electrons, and neutrons for specific atoms.
Pearson
The Chemical Context of Life
An educational presentation includes atoms, molecules, the four major elements, as well as neutrons and protons. Additionally, slides focus on atomic number, mass number, atomic weight, polar and nonpolar covalent bonding, ionic...
Curated OER
Matter Overview
There is not much information in this presentation. It serves more as a guide to direct the flow of your introductory chemistry lecture. Each slide displays a large title and a smaller phrase meant to explain it. For example, one title...
Virginia Department of Education
A Crystal Lab
Young chemists grow ionic crystals, metallic crystals, and supersaturated crystals in three different lab experiments. Observing these under a microscope allows pupils to compare the various structures.
Curated OER
How Do Atoms Stick Together?
For this chemical bonding worksheet, students answer 76 questions about compounds, Lewis dot structures, intermolecular forces between atoms, electronegativity and bonding and types of bonds.
Curated OER
Naming and Covalent Compounds
In this naming and covalent compounds learning exercise, students name 12 compounds using a chart of polyatomic ions if needed. They also draw covalent bonds in 6 compounds and answer 6 questions about ionic bonds, covalent bonds and...
Curated OER
Introduction to Bonding
In this chemistry worksheet, students create new and true statements while replacing false ones in order to properly define parts of the concept of bonding.
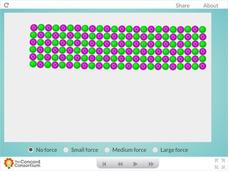
Concord Consortium
Ceramic Forces
Why are bricks more likely to break than bend? Young science scholars peer inside a ceramic block and examine the effects of downward force at the molecular level. Learners can apply three different levels of force before observing their...
Curated OER
Common Ions and Their Charges
In this chemistry activity, students can use the table to observe and name the common types of ions and determine the charges to explain the gain or loss of electrons.
Curated OER
Naming and Covalent Compounds
In this compounds worksheet, students practice naming compounds and classifying them as ionic, covalent, or polyatomic compounds. This worksheet has 12 fill in the blank and 12 problems to solve.
Curated OER
Naming Compounds
In this naming compounds worksheet, students are given a chart to determine if the compound they are naming is ionic, covalent or polyatomic. Students practice identifying and naming ionic, covalent and polyatomic compounds. They define...
Curated OER
Writing Chemical Formulas
Learners study how to write chemical fomulas by reviewing the combinations atoms form into compounds. They write a procedure to test various substances and name the compounds and write formulas. As they construct models for formulas and...
Curated OER
Animated Chemical Bonding
Students demonstrate their understanding of a chemical process. In this exploratory lesson students create a clay animation that shows how a specific type of chemical bond.