Curated OER
Atoms and Matter
In this states of matter worksheet, students complete an 8 question multiple choice online interactive quiz about matter and atoms. The quiz can also be printed out and completed on paper.
Curated OER
Conversion to Moles, Grams, and Atoms Worksheet
In this chemistry conversion worksheet, students convert to moles, grams, and atoms as they respond to 5 short answer questions.
Curated OER
Calculating the Number of Sub-Atomic Particles in an Atom
In this atoms instructional activity, learners compare the position, relative charge and relative mass of sub-atomic particles. Students define mass number and atomic number. This instructional activity has 2 graphic organizers and 4...
Curated OER
Ionic bonding
Students explore ionic bonding. They draw examples of ionic bonding and explain the activities of the electrons of the elements. Students use paper plates and candy to draw electron configurations of given atoms.
Curated OER
Anatomy of an Element
Learners discuss matter and atoms using this resource. First, they look at a website describing atoms. Then, they learn about the periodic table and discuss how it is organized. Finally, they create a comic strip to display their...
Curated OER
Mighty Molecules
Students examine a PowerPoint production on molecules. Then they construct their own molecule and define what an atom, molecule, and matter is. They make a H2, HCI, and an H2O molecule. Finally, a drawing to accompany the actual model is...
Curated OER
The Elements
Fifth graders explore the differences between elements. In this elements and atoms lesson students complete an activity that shows how chemicals react to a flame test.
Curated OER
Physical Science Project-Atom
Fifth graders investigate atoms. In this atom lesson, 5th graders investigate the parts of an atom. Students explore atom numbers and determine the number of electrons is in each atom.
It's About Time
Are Atoms Invisible?
Wow, an experiment that allows the class to participate in a missile war! Pupils discuss Thomsons's theory of cathode rays and simulate Rutherford's historical experiment to learn about atomic structure. They conclude this fourth lesson...
Concord Consortium
Molecular View of a Liquid
Pour on the knowledge with an engaging states of matter interactive! Young physical scientists view the atoms within a liquid to explain what gives a liquid its unique properties. The resource allows users to highlight and observe two...
PHET
Atomic Interactions
Who knew atoms could be attracted to each other? A simulation allows learners to see how forces between atoms influence the attraction between them. Classes see the attractive and repulsive forces adjusting as distance between atoms...
Concord Consortium
Understanding Probability Maps
What's the likelihood of your class understanding probability maps? Young scientists explore the concept using a virtual dartboard in an interesting simulation. The darts establish a pattern around the bull's-eye, which provides a visual...
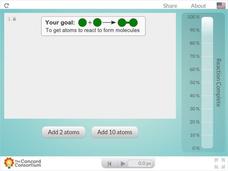
Concord Consortium
Concentration and Reaction Rate
Does concentration affect the rate of a chemical reaction? Science scholars observe and control a chemical reaction in an interactive simulation. The resource allows pupils to add atoms to the reaction vessel and monitor the reaction's...
National Nanotechnology Infrastructure Network
Sometimes We Need Large Numbers to Describe Small Things
String the class along to help them understand large numbers. A multi-disciplinary lesson uses literature, science, and math to look at very large numbers. Pupils conceptualize how much a million is using literature before performing a...
Concord Consortium
Stoichiometry and Balancing Equations
Is your stoichiometry lesson plan just not adding up? Incorporate an exciting interactive to balance things out! Chemistry scholars manipulate the number of molecules added to the reaction vessel, then observe as bonds form and break as...
American Chemical Society
The Same Parts Can Make Many Objects
Snap to it! Young experimenters use Snap Cubes to model how a larger object is created from a smaller parts. Using a guide, they rearrange their cubes to create all possible arrangements of four and five cubes.
Curated OER
Energy and Atoms
In this atoms worksheet, students review atoms, atomic number, and mass number. Students determine what happens when atoms become radioactive. This worksheet has 8 fill in the blank and 9 short answer questions.
Curated OER
Atom Models Through the Ages
Eighth graders explain the development of the atomic model over time. In this chemistry lesson plan, 8th graders research about the scientists who made a contribution to the atomic theory. They create a three-dimensional model based on...
Curated OER
What's the Matter? Locating Electrons in an Atom
Students roll dice in order to simulate the probability of locating an electron in a certain region around the nucleus.
Curated OER
Chemistry: Atoms
In this chemistry worksheet students complete a series of multiple choice and short answer questions on elements, chemical reactions and balancing equations.
Curated OER
Atoms and Chemical Bonding
An online resource, this worksheet has your chemistry learners complete four data tables with information about atoms and chemical bonds. The first table addresses atomic number and mass, as well as numbers of protons, neutrons, and...
It's About Time
How Atoms Interact with Each Other
Connect the dots and assist young chemists as they demonstrate covalent and ionic bonding. Class members use their knowledge of valence electrons to predict compound formulas as they arrange electrons into various bonding structures to...
Texas State Energy Conservation Office
Investigation: Splitting Atoms
In a simple activity, physical scientists model nuclear fission using a droplet of oil. This can be used alone in a unit on different types of energy, or as part of the energy conservation unit produced by the Texas State Energy...
Concord Consortium
Non-Bonding
What makes helium so Noble, anyway? Explore the bonding properties of helium versus those of hydrogen using an interactive resource. Science scholars manipulate atoms of each element and observe changes in potential energy as the atoms...