Hi, what do you want to do?
Professor Dave Explains
The Common Ion Effect
We've learned a few applications of the solubility product, so let's learn one more! This is called the common ion effect, and it can be a good way to influence the solubility of a particular ionic compound in solution, or even promote...
Professor Dave Explains
Solubility Product Constant (Ksp)
We've learned that some ionic solids are totally water insoluble, but in fact this is a slight oversimplification. Even such solids will dissolve to some minuscule degree, and we can measure this, and do calculations with it. Let's learn...
JFR Science
Introduction to Solutions: Solutions and Concentration
Don't break your concentration when learning about solutions. Pupils watch a video in the JFR Science series that explains what solutions are and how to calculate solution concentrations. The resource covers solubility and molar...
Khan Academy
Khan Academy: Solubility and Solubility Product Constant
Introduction to solubility and solubility constant with an example of solubility calculation. [11:45]
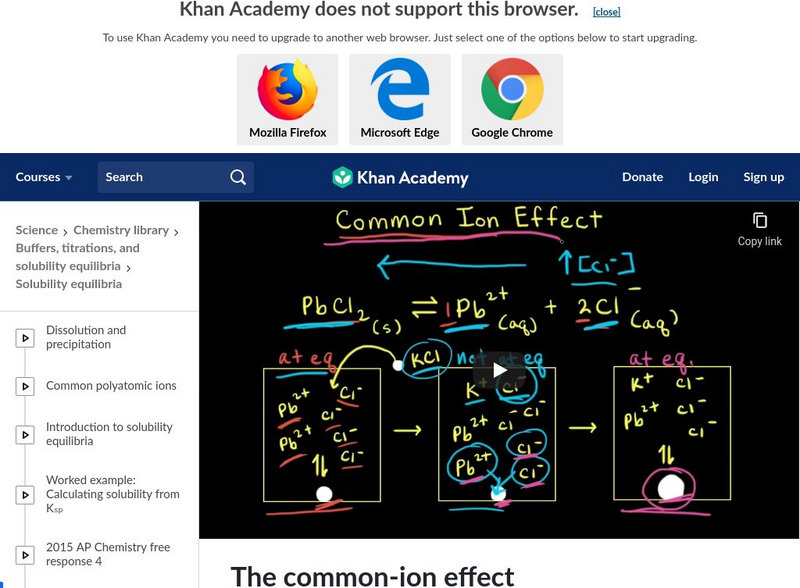
Khan Academy
Khan Academy: Solubility and the Common Ion Effect
Using an ICE table to calculate molar solubility. [8:15]
Khan Academy
Khan Academy: Solubility and Complex Ion Formation
An explanation of calculating the new equilibrium constant to help understand how complex ions are formed. [14:49]
Khan Academy
Khan Academy: 2015 Ap Chemistry Free Response 4
The video reviews over a free response from an AP exam where students were asked to calculate the molar solubility of calcium hydroxide in a 0.10 M calcium nitrate solution [9:19]